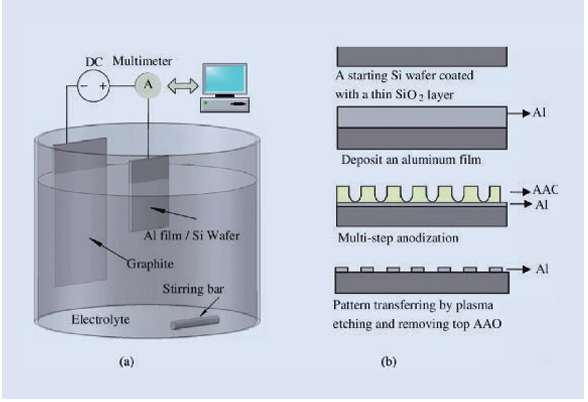
The simplest way to understand how anodizing works is in the name. To anodize a part, you connect it to a positive terminal of an electrical circuit, also called an anode. You then submerge the part in an acidic electrolyte bath solution. This solution contains chemical compounds, such as sodium phosphate, which fill the bath with positive and negative ions.
Once the part is submerged and secured to a hanger so it doesn’t move around, you apply the negative end of the circuit, or cathode, to a metal electrode in the bath. When you send voltage through the circuit, the negative electrode attracts positive ions (cations) from the part, and the aluminum part attracts negative O2 ions (anions) from the solution.
When positive aluminum ions leave the part’s surface, it becomes porous, reacting with the negative O2 ions to grow a layer of aluminum oxide.